CAMPBELL, Calif., April 21, 2026 – Saama, a provider of AI-based solutions that accelerates clinical development and commercialization, has been named a Luminary in the “Innovation Watch: AI Applications in Clinical Development 2026” by Everest Group. The annual report evaluated 18 leading AI solution providers and features Saama within the top category of Luminaries for its strength in market performance and ecosystem drivers, including market adoption, solution maturity, partnerships, and investments.
The 2026 report assessed Generative AI, Agentic AI, and advanced AI solutions applied to clinical development use cases, including study start-up, data management and biostatistics, patient recruitment and engagement, safety and pharmacovigilance, and regulatory submissions.
“Through exclusive data partnerships, Saama enhances the performance and scalability of its AI models across clinical data environments,” said Nisarg Shah, Practice Director at Everest Group. “Saama’s investments in innovation demonstrate both technical depth and execution maturity, complemented by strong market positioning through thought leadership, AI training initiatives, and active ecosystem engagement. These capabilities reinforce Saama’s standing as a Luminary in AI-enabled clinical analytics.”
The Saama platform of AI-enabled SaaS solutions is delivering value today by supporting the full spectrum of clinical development. Efficiencies across clinical research studies include:
- 35% reduction in time to data discovery with Data Hub
- 40% time savings for patient data review with Patient Insights
- 20,000+ hours of manual work saved using Smart Data Quality (SDQ)
- 60-70% reduction in manual analysis time compared to traditional analysis using TLF Analyzer
- 30% reduction in content drafting time with AI-powered Document Generator
“This recognition from Everest Group reflects something we hear directly from our customers every day, with 100s of studies scaled on the platform & putting humans firmly in control, Saama offers a proven ecosystem that harnesses the power of defensible & responsible AI-driven insights. This is making a tangible difference in how clinical trials are designed, executed, and completed from Protocol through Submissions,” said Ari Srinivasan, Chief Customer Success and Growth Officer, Saama. “Being named a Luminary reinforces our commitment to building solutions that Sponsors and CROs can rely on to bring life-changing therapies to patients sooner.”
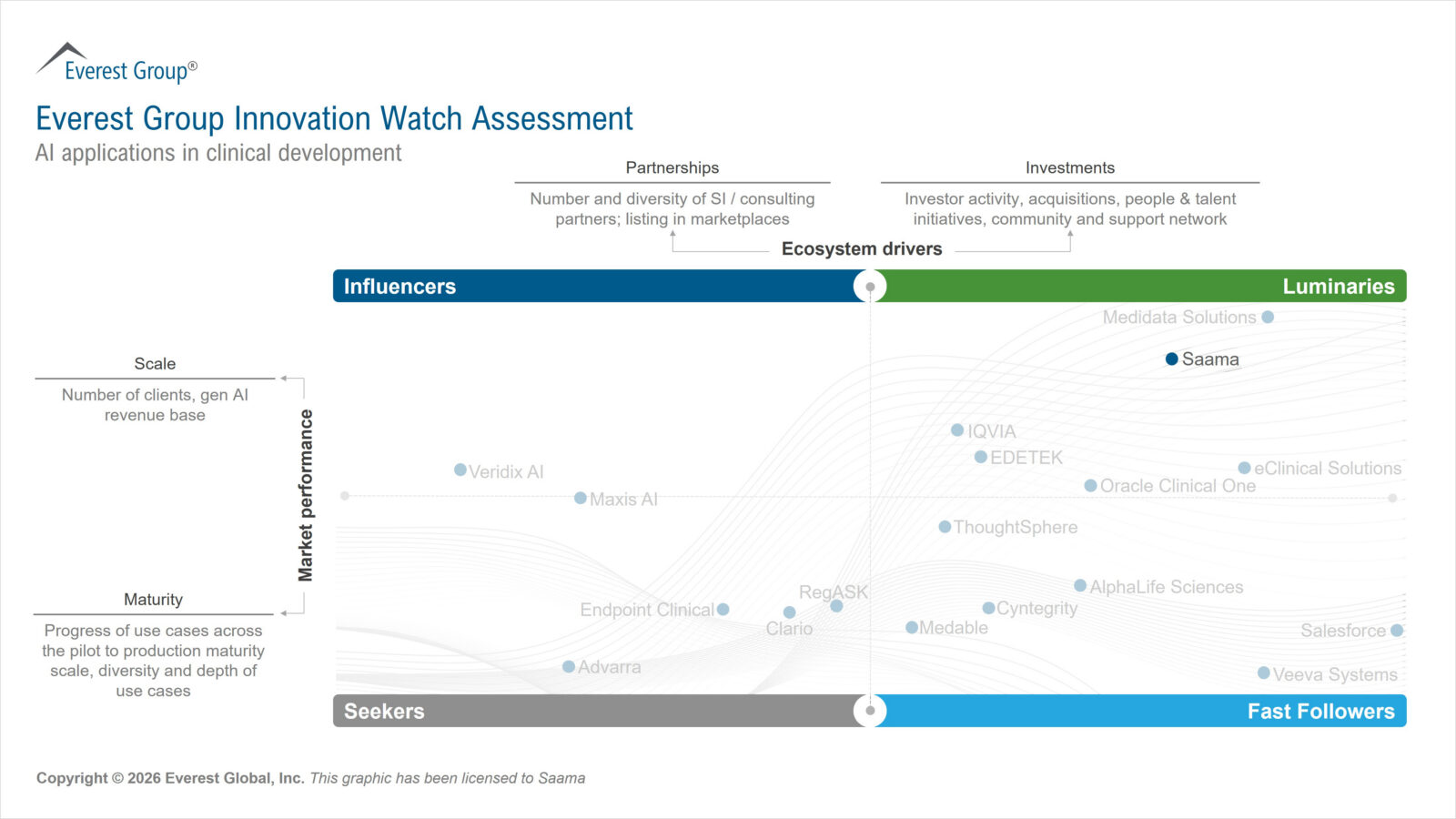
Saama’s full Innovation Watch Assessment is available for download.
To learn more about the Saama platform, its custom solutions and services, or to schedule a personalized demonstration, visit saama.com.
About Saama
Saama develops healthcare and life science products and solutions that accelerate the delivery of therapies to patients. Saama also provides specialized data, analytics, and AI services to other industries. The company’s AI-driven clinical data management platform enables pharma and biotech companies to streamline clinical and commercial operations, improve data quality and access, and gain faster insights across their clinical trials. The Saama platform powered the clinical trial that led to the world’s first COVID-19 vaccine. Headquartered in Campbell, CA, with employees around the globe, Saama is committed to helping customers save and improve lives. Discover more at saama.com.
Contact:
Heather Shea
Catalytic Agency on behalf of Saama
[email protected]
